Argon is important for OES Spectrometers analyzing light elements such as Carbon, Phosphorous, Sulfur, and Nitrogen. These elements are below 200 nanometers (nm) in wavelength and the argon allows the optic of a spectrometer to see from 200 and below. Issues with argon cause a number of problems for a spectrometer, most importantly, the false rejection of material that is otherwise good.
There are a few common issues associated with the argon system of an optical emissions spectrometer.
Argon Purity
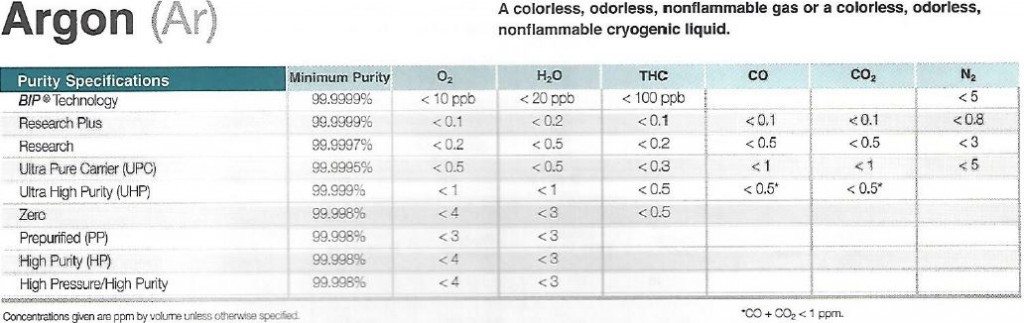
OES Spectrometer users assume argon is argon but there are grade differences. 4.8 argon is 99.998 percent pure argon while 5.0 is 99.999% pure argon. The slight jump in purity is the result of a number of factors all of which contribute to 5.0-grade argon being the bare minimum for use in a spectrometer.
Jackson Welding Supply of Pittsburgh, PA provides a list of available argon purity levels and trade names. Grade 4.8 goes by the trade name Zero while grade 5.0 goes by Ultra High Purity or UHP for short. Below is a list of trade names, purity specifications and ppm limits of trace elements contained within the tank.
Argon Purity Grades
Grade 5.0 Argon is treated differently than 4.8-grade argon. Every time a bottle of 5.0 argon is depleted the supplier cleans the interior of the bottle to remove CO2 and H2O vapors. Some bottles also contain a BIP (Built-In Purifier) device to scrub the argon before it leaves the bottle. These processes and devices mean that the argon is not carrying contaminates with it upon exiting the bottle.
Argon 4.8 vs 5.0
Grades 4.8 and below do not experience this cleaning process or incorporate a BPI device. Many times H2O vapor and CO2 gas get mixed in with the argon. For welders, the slight mixture of H2O and CO2 isn’t a problem but for spectrometer operators trying to determine Carbon, Boron or Nitrogen it’s a real issue. The oxygen present in H2O and CO2 destroys wavelengths from 200 nm and below. This is why the incorrect grade of argon will show operators inaccurate information.
Importance of Argon Pressure
The amount of pressure within an argon bottle is important for maintaining proper operation. When the pressure reaches 500 psi it is time to place an order for a new bottle; when it reaches 300 psi it’s time to disconnect the bottle and connect a new bottle. 300 psi is considered empty because any lower pressure will cause the argon to ripple through the line instead of flowing, and any CO2 or H2O trapped at the bottom of the cylinder will move with the argon.
OES Spectrometer Argon Tubing
There are two types of tubing on an OES Spectrometer; argon feed tubing and exhaust tubing. The feed tubing is 6mm in diameter for mobile and laboratory units and transports argon from the pressurized bottle to the sample area. The exhaust tubing removes the argon and any soot from the sample area. The exhaust tubing for a mobile unit is 6mm in diameter, but the exhaust tubing for a laboratory unit is 12mm in diameter.
Argon Feed Tubing
Argon-grade tubing is made from polypropylene. Polypropylene is used because the tubing isn’t porous. Porous tubing allows argon to fill the voids and, once moved from the void, pick up particles that contaminate the optic.
Leaks in the tubing allow outside gases to enter the line and contaminate the system. It’s a commonly held belief that pressurized argon gas will leave through cracks but the outside matter will not enter; this is simply not true. Due to the Venturi effect, outside atmospheric gas will get into the system and mix with the argon.
Argon Exhaust Tubing
Exhaust tubing is important as well because it provides back pressure in the system. When an exhaust line becomes clogged, debris from the sample test area is no longer leaving the test stand. Argon feed tubing free of leaks and/or an unobstructed exhaust line work together to move argon from the pressurized bottle through the sample stand to an exit point without impacting the test results.
Flushing the System
The biggest reason for callbacks or Service Shop Technical Support regarding instrument questions is caused by shortening the argon flushing time. Mobile OES users frequently experience this problem. 45 minutes to 1 hour is the recommended duration for the argon system to flush. Many users will flush for 10-20 minutes, then test a sample and receive an inaccurate reading. 45-60 minutes allows enough time for the argon to remove particles and atmospheric gases from the system.
Are You Paying Attention to Argon
Grade, argon delivery, pressure, and flush duration all play an important role within the argon system and support low levels of analysis. If one of these components isn’t in prime working condition, the entire system will fail and hurt operational performance. For a review of your spectrometers argon system or in-house operator training, contact Verichek Technical Services, Inc.